How to Read the Marks on an Iv Bag
Chapter eight. Intravenous Therapy
8.4 Priming IV Tubing and Changing IV Fluids and Tubing
Primary and secondary Four tubing and addition devices (extension tubing) must exist primed with Four solution to remove air from the tubing. Priming refers to placing 4 fluid in Four tubing to remove all air prior to attaching the IV tube to the patient. 4 tubing is primed to prevent air from inbound the circulatory system. An air embolism is a potential complication of Iv therapy and can enter a patient's blood system through cut tubing, unprimed Iv tubing, admission ports, and drip chambers with too trivial fluid (Perry et al., 2014). It is unknown how much air will cause death, only deaths accept been reported with as picayune as 10 ml of air. The best style to avoid air bubbles in 4 tubing is to forbid them in the first place (Perry et al., 2014). New IV tubing may also be required if leaking occurs effectually the tube connecting to the 4 solution, if the tubing becomes damaged, or if it becomes contaminated. Checklist 66 outlines the procedure of priming Iv tubing.
Disclaimer: Always review and follow your hospital policy regarding this specific skill. | ||||
Safety considerations:
| ||||
Steps | Additional Information | |||
| ane. Perform manus hygiene. | This step prevents the manual of microorganisms. | |||
| ii. Check society to verify solution, rate, and frequency. | This ensures IV solution is correct and helps forestall medication fault. | |||
| 3. Assemble supplies. | You will need IV solution, main IV tubing, fourth dimension label, change label, booze swab, and basin or sink.  | |||
| 4. Remove IV solution from outer packaging and gently squeeze. | Tear the perforated corner of the outer packaging; bank check colour, clarity, and expiration engagement.  | |||
| 5. Remove primary IV tubing from outer packaging. |  | |||
| half dozen. Motion the roller clamp nearly iii cm below the baste sleeping room and close the clamp. | 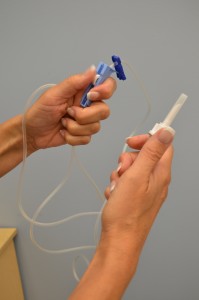 | |||
| 7. Remove the protective comprehend on the IV solution port and keep sterile. Remove the protective cover on the IV tubing fasten. | Be careful and do non contaminate the spike.  | |||
| 8. Without contaminating the solution port, carefully insert the IV tubing spike into the port, gently pushing and twisting. | 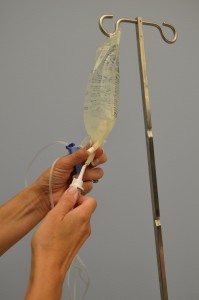 | |||
| nine. Hang bag on 4 pole. | The Iv handbag should be approximately one metre to a higher place the Iv insertion site. | |||
| 10. Make full the drip bedroom one-tertiary to one-half full past gently squeezing the chamber. Remove protective cover on the end of the tubing and continue sterile. | Filling the drip chamber prevents air from entering the IV tubing. 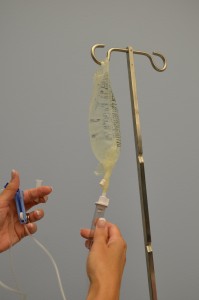 | |||
| xi. With distal end of tubing over a bowl or sink, slowly open up roller clamp to prime the Iv tubing. Invert backcheck valve and ports equally the fluid passes through the tubing. Tap gently to remove air and to fill with fluid. | Inverting and tapping the access ports and backcheck valve helps displace and remove air when priming the 4 tubing. 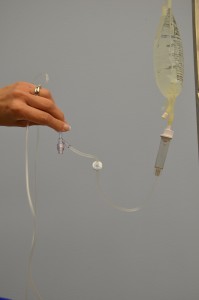 | |||
| 12. In one case Four tubing is primed, check the entire length of tubing to ensure no air bubbles are present. | This pace confirms that air is out of the IV tubing. | |||
| 13. Close roller clench. Cover terminate with sterile expressionless-ender or sterile protective cover. Hang tubing on IV pole to prevent from touching the ground. | Go on the distal terminate sterile prior to connecting IV to patient. | |||
| xiv. Characterization tubing and IV bag with date, time, and initials. | Label 4 solution bag equally per agency policy. Do not write direct on the 4 handbag. 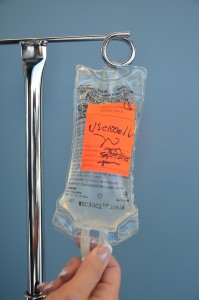 | |||
| fifteen. Perform hand hygiene. | This reduces the transmission of microorganisms.  | |||
| Data source: Fulcher & Frazier, 2007; Perry et al., 2014. | ||||
Video eight.ane
Lookout the videoPriming Four Lines by Renée Anderson & Wendy McKenzie, Thompson Rivers University.
IV solutions are considered sterile for 24 hours. An IV solution may exist changed if the medico's order changes, if an IV solution infusing at TKVO is expired after 24 hours, or if the Four solution becomes contaminated. To alter an 4 solution bag, follow Checklist 67.
Disclaimer: Always review and follow your hospital policy regarding this specific skill. | ||||
Steps | Additional Information | |||
| i. Verify and select correct Four solution bag and compare to the medication administration tape (MAR) or dr. orders. | IV solutions are considered a medication and must be checked using the SEVEN RIGHTS x 3, every bit per agency policy.  | |||
| 2. Innovate yourself, identify patient, and explain process. | Proper identification of a patient prevents medication errors. Explaining the procedure provides an opportunity for the patient to ask questions. | |||
| 3. Perform hand hygiene. | Hand hygiene prevents the transmission of microorganisms. | |||
| 4. Remove outer plastic packaging and clasp bag to test for leaks and expiration appointment. Assess for precipitates or cloudiness. Hang new IV solution on Iv pole. | This ensures the correct IV solution is used.  | |||
| 5. Interruption the EID or close the roller clench on a gravity infusion set. | Stops the infusion to forestall air bubbling from forming in Four tubing. | |||
| 6. Remove protective plastic cover from the new IV solution tubing port. | Proceed IV tubing port sterile at all times. If Four tubing port becomes contaminated, dispose of it immediately and supercede. | |||
| seven. Remove the old Four solution bag from the IV pole. Turn IV bag upside downward, grasping the tubing port. With a twisting motility, carefully remove Four tubing spike from sometime Iv solution bag. | Removing old solution from IV pole prevents spilling of solution. Ensure IV tubing spike remains sterile during removal to avert contaminating IV tubing. | |||
| eight. Using a gentle twisting motion, firmly insert the fasten into the new IV bag. | This ensures that a sterile technique is used during the process. 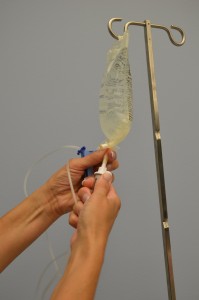 | |||
| ix. Fill the drip chamber by compressing information technology between your thumb and forefinger. Ensure the baste chamber is one-third to one-half total. Check 4 tubing for air bubbles. | Fluid in the drip chamber helps foreclose air from existence introduced into IV tubing. 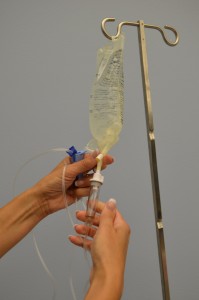 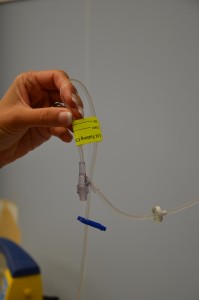 | |||
| 10. Open clamp and regulate IV infusion rate via gravity, or press start on the EID as per md orders. | Once rate is prepare, count the drops per infinitesimal on the gravity set or ensure the EID is running at the correct rate as per doctor orders. 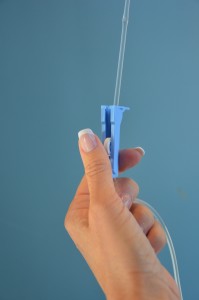 | |||
| eleven. Label new Iv solution bag equally per bureau policy. Time tape gravity IV solutions as per agency policy | Labelling IV solutions provides piece of cake viewing of infusing solutions and additives.  | |||
| 12. Dispose of used supplies, perform hand hygiene, and document IV solution handbag change according to bureau policy. | Document time, date, blazon of solution, rate, and full volume. | |||
| Data source: Fulcher & Frazier, 2007; Perry et al., 2014. | ||||
Video 8.2
Watch the video Irresolute Four bagsby Renée Anderson & Wendy McKenzie, Thompson Rivers Academy.
Checklist 68 describes how to change the Iv tubing assistants prepare and IV solution at the aforementioned time.
Disclaimer: Always review and follow your hospital policy regarding this specific skill. | ||||
Steps | Additional Data | |||
| ane. Verify md orders for the type of solution, rate, and duration. Collect necessary supplies. | This pace verifies the patient's need for IV fluids/medications. It also confirms the right rate and solution for patient safety. | |||
| ii. Perform hand hygiene. | Hand hygiene prevents the transmission of microorganisms. | |||
| 3. Identify yourself, identify the patient using two identifiers, and explain the procedure to the patient. | Proper identification of patient prevents errors. 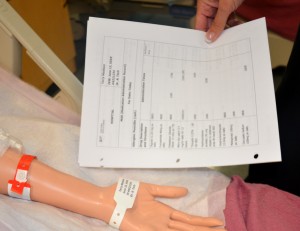 | |||
| 4. Prime number new administration prepare using a new 4 solution purse and new IV tubing. | Four solutions are considered a medication. Prime as per Checklist 66. Keep distal protective cap fastened to IV tubing to ensure sterility of distal end. Label Iv solution and 4 tubing as per agency policy.  | |||
| 5. Hang new assistants set (primed primary line and 4 solution) on 4 pole. | This prepares the equipment and adheres to the principles of hygienic technique. | |||
| 6. Clamp old 4 administration gear up. Remove Four tubing if on an EID. | Cease the flow of infusion during tubing and solution change. | |||
| 7. Clean the connectedness between the distal end of old Four tubing and the positive pressure cap. Scrub the surface area for fifteen seconds and permit it dry out for thirty seconds. | Proper disinfection of equipment decreases bacterial load and prevents infections. 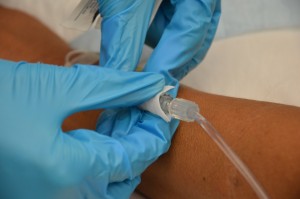 | |||
| 8. Remove the protective cap on the distal end of the new IV administration prepare. |  | |||
| 9. Advisedly disconnect the sometime tubing from the positive pressure level cap and insert the new 4 tubing into the positive pressure cap attached to the extension tubing. | 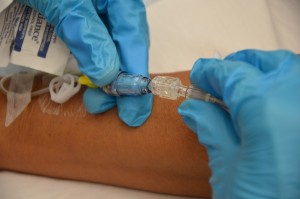 | |||
| 10. Open the roller clamp on the new tubing to regulate menstruation rate, or insert new tubing into the EID and restart 4 rate. | This step ensures the IV solution is infusing at the correct rate.  | |||
| xi. Check Four site for patency, and signs and symptoms of phlebitis. | IV site should be costless from redness, swelling, and pain. Dressing on 4 site should exist dry out and intact. 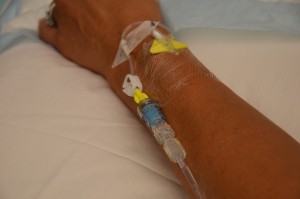 | |||
| 12. Discard quondam supplies and perform hand hygiene. | This step prevents the spread of microorganisms.  | |||
| xiii. Certificate procedure as per agency policy. | Certificate the engagement and time of Four tubing and solution change. | |||
| Information source: BCIT, 2015b; Fulcher & Frazier, 2007; Perry et al., 2014 | ||||
- How long can IV solution be used?
- What is the purpose of removing air from Iv tubing?
Source: https://opentextbc.ca/clinicalskills/chapter/8-4-iv-assessment-maintenance-troubleshooting-and-discontinuation/
ارسال یک نظر for "How to Read the Marks on an Iv Bag"